Diamond:
a. Diamond is an allotrope of carbon. It is used in jewellery and is the most precious crystal.
b. Covalent bonds between sp3 hybridized carbon atoms continue in all directions to form a giant network.
c. Diamond is very strong and hard due to presence of strong covalent bonds.
d. It is the hardest material and has very high melting point (355°C).
Graphite:
a. Graphite is an allotrope of carbon in which each carbon atom is sp2 hybridized and forms covalent bonds with three other sp2 hybridised carbon atoms. This results in formation of interlinked six membered rings.
b. π bonds are formed between carbon atoms by utilizing half filled unhybridised 2pz orbital on each carbon atom. Layers of carbon atoms are formed. 
c. Electrons present in delocalized molecular orbitals can move freely and conduct electricity. Hence graphite is a good conductor of electricity.
d. Graphite is sufficiently hard due to presence of relatively stronger covalent bonds. The adjacent layers of carbon atoms are held together by weak van der Waal’s forces of attraction.
e. The layers of carbon atoms in graphite can slide over other layers. Hence, graphite is used as lubricant for reducing friction.
f. It is used in ribbons used for printing in computers and typewriters. It is also used in lead pencils.
Fullerene:
a. Fullerene is an allotrope of carbon. It is also called Buckball or Buckminster fullerene.
b. When a high power laser was focused on carbon, fullerene was formed. It has formula C60 and shape of a soccer ball (hollow sphere).
c. sp2 hybridized carbon atoms occupy sixty equidistant places on this sphere. They form hexagons and pentagons.
d. Fullerenes are present in carbon soot. 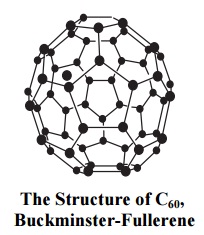
e. Fullerene reacts with potassium to form K35C60. This compound is a superconductor of electricity at 18 K. It reacts with transition metal to form a catalyst.
f. Tubes made from fullerene and graphite are called nanotubes. These are used as high strength materials, electric conductors, molecular sensors and semiconductors.
